 
(ed.), Gas Phase Ion Chemistry, vol. 2, New York: Academic Press, p. 53. (1979), "Electron affinities", in Bowers, M. NIST chemistry webbook after Borshchevskii et al. The electron affinities of the radicals OH and SH are the most precisely known of all molecular electron affinities. Many more have been listed by Rienstra-Kiracofe et al. The electron affinities E ea of some molecules are given in the table below, from the lightest to the heaviest. They may have lifetimes of the order of microseconds to milliseconds, and invariably autodetach after some time. Negative ions formed in these cases are always unstable. Conversely electron removal from the anion formed in this way releases energy, which is carried out by the freed electron as kinetic energy. when capture can occur only if the impinging electron has a kinetic energy large enough to excite a resonance of the atom-plus-electron system. Negative electron affinities can be used in those cases where electron capture requires energy, i.e. The latter can be regarded as the ionization energy of the –1 ion or the zeroth ionization energy. 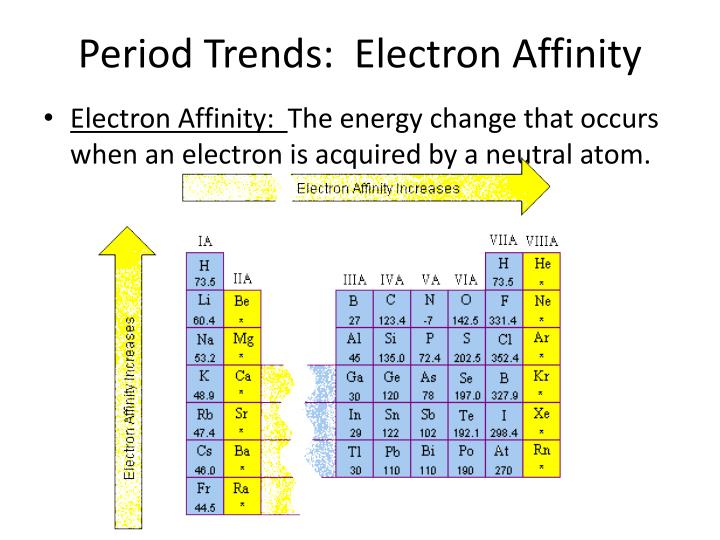
The second (reverse) definition is that electron affinity is the energy required to remove an electron from a singly charged gaseous negative ion. First, as the energy that is released by adding an electron to an isolated gaseous atom. Electron affinity can be defined in two equivalent ways. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
